Psychiatric Diseases in Pregnancy
- In March 2019, the U.S. Food and Drug Administration approved Zulresso (Brexanolone) injection for intravenous (IV) use for the treatment of postpartum depression (PPD) in adult women. This is the first drug approved by the FDA specifically for treatment of PPD and is currently available only under a restricted distribution program due to concerns about serious risks including excessive sedation or sudden loss of consciousness during administration. Specific restrictions and requirements are planned to be detailed on the medication’s associated black box warning label.
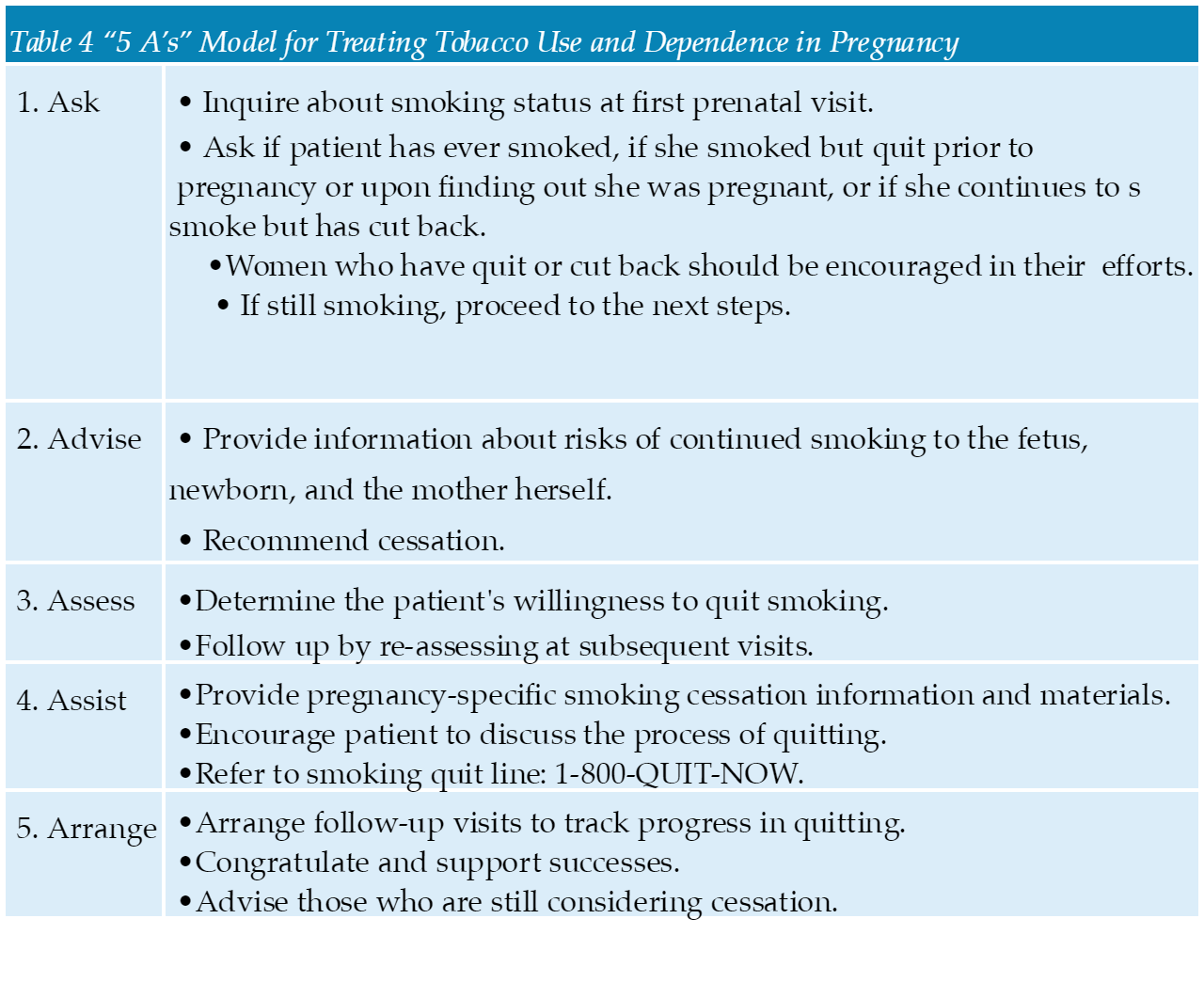
- All women should be screened multiple times during pregnancy and in the postpartum period for depression, regardless of their risk factors or medical history. Many tools such as the Edinburgh Postnatal Depression Scale form are available to assist in screening.
- When considering intrapartum pain management in women with opioid use disorder, it is important to avoid drugs with an opioid antagonist component, such as nalbuphine, as this can precipitate withdrawal symptoms. Use of other medications such as NSAIDs can reduce the amount of opioids required.
- The effect of marijuana use in pregnancy is unclear, as studies are confounded due to co-existing mental health disorders, socioeconomic status, parental education, and cigarette smoking. Despite these limitations, several studies have demonstrated an association with preterm labor, low birth weight, NICU admission, and stillbirth. As with any drug of abuse, abstinence during pregnancy should be encouraged.


.png)







